
Daltons Atomic Theory (1803) British chemist John Dalton proposed that matter is composed of indivisible, indestructible tiny particlesatoms. This resource is part of the Atoms and the Periodic Table BundleThe Atomic Theory Timeline Project is everything you need to give your chemistry students a nice background on the history of the atom Students use skills of synthesizing. When talking about the atomic model timeline project, the Scientists understanding of atoms has gone through several stages, and the following are five main historical models. Dalton’s theory laid the foundation for our understanding of chemical reactions and the conservation of mass. Atomic Theory Timeline Project: A Visual History of the Atom. He proposed that atoms were tiny, indivisible particles that combined in fixed ratios to form compounds. For example, copper and chlorine can form a green, crystalline solid with a mass ratio of 0.558 g chlorine to 1 g copper, as well as a brown crystalline solid with a mass ratio of 1.116 g chlorine to 1 g copper. In the early 19th century, John Dalton introduced his atomic theory. 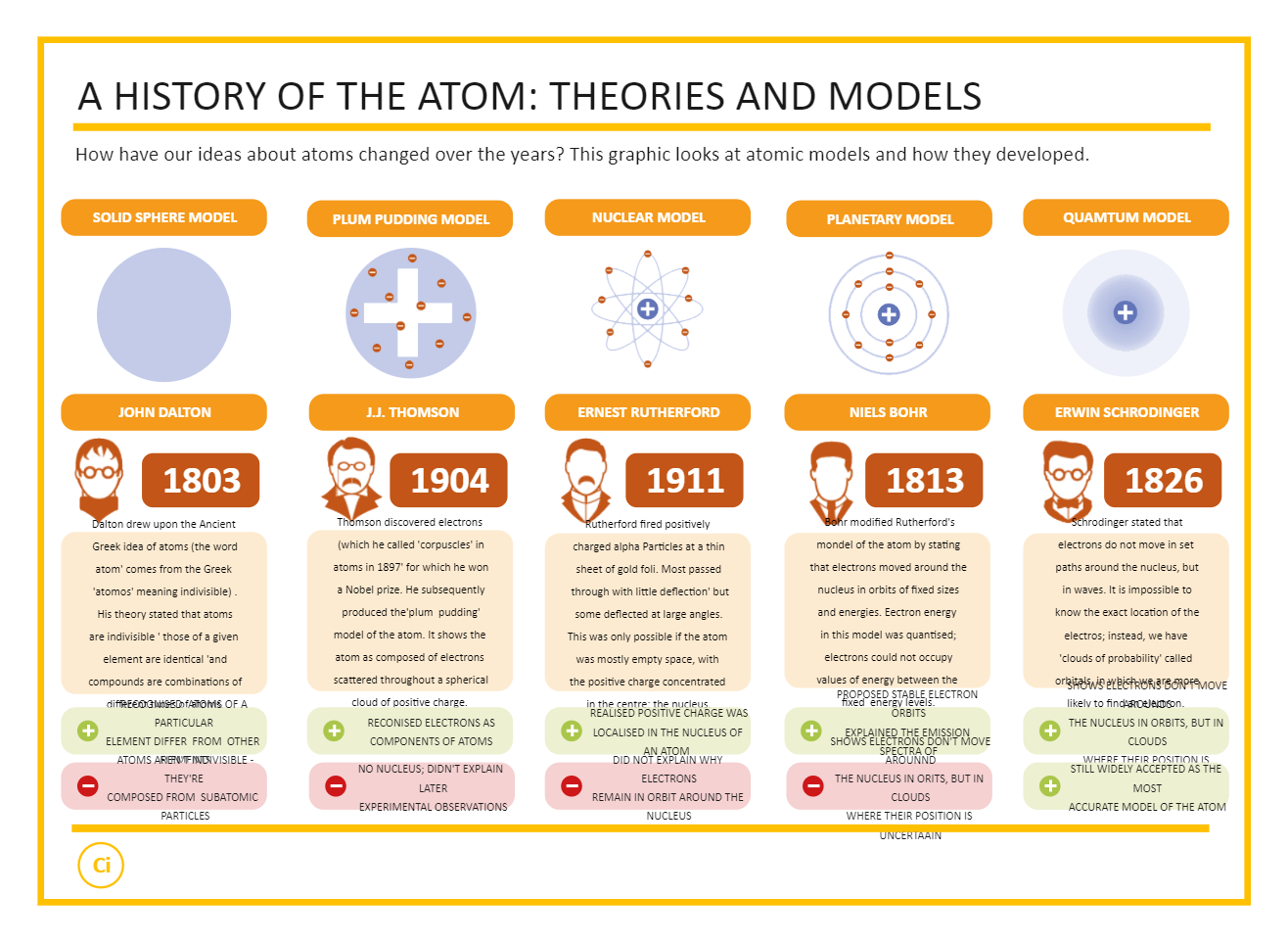
The law of multiple proportions states that when two elements react to form more than one compound, a fixed mass of one element will react with masses of the other element in a ratio of small, whole numbers. In this study guide, you can revise how the periodic table arranges elements according to. This introduction to chemistry, from a practical discipline in ancient times to the science it is today, touches on both major advances and discarded theories. He believed all materials on earth were made up of small amounts of each element. Unit 1 Matter and the Rise of Atomic Theory: The Art of the Meticulous. Instead he proposed that all things are made up of the four elements fire, water, air, and earth. Aristotle did not believe in the atomic theory. A chronological overview of the key discoveries and models of the atomic theory from ancient to modern times, with references and diagrams. For example, there are many compounds other than isooctane that also have a carbon-to-hydrogen mass ratio of 5.33:1.00.ĭalton also used data from Proust, as well as results from his own experiments, to formulate another interesting law. Atoms are made from protons, neutrons and electrons. His beliefs of the atom contradicted his teachers beliefs. That is, samples that have the same mass ratio are not necessarily the same substance. It is worth noting that although all samples of a particular compound have the same mass ratio, the converse is not true in general.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
